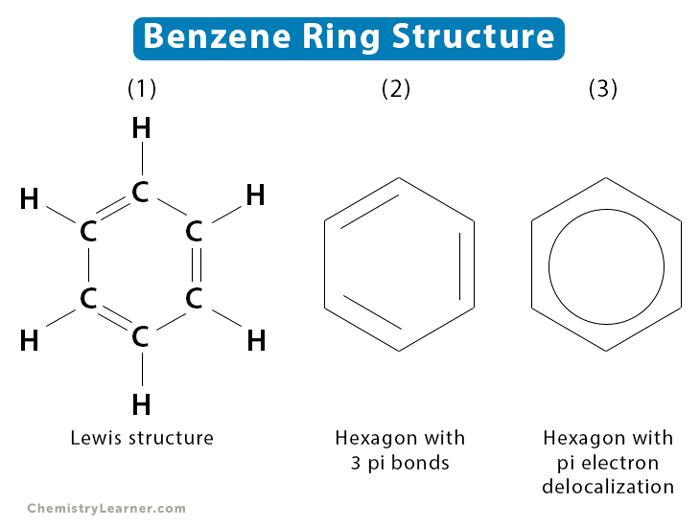
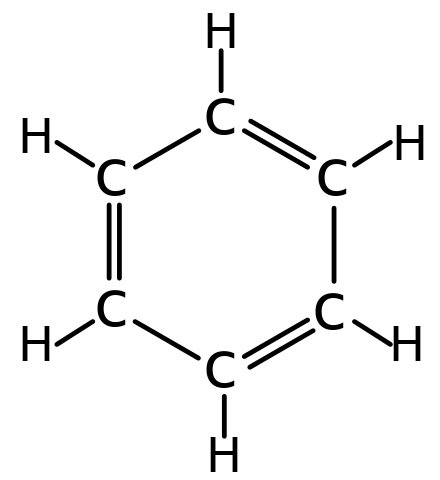
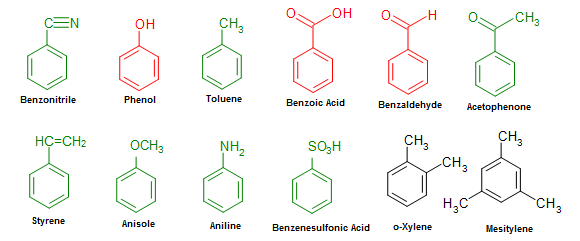
Aromatic ring arrangement categories. Phenyl rings demonstrating the... | Download Scientific Diagram

Why do we not add “benzene” to the answer in the image attached since there is also a benzene ring and benzene( or phenyl in cases where it is considered to be
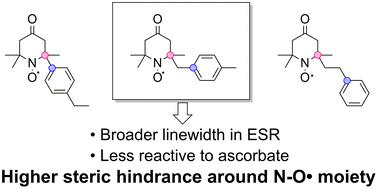
Substituent effects of the phenyl ring at different positions from the α-carbon of TEMPO-type nitroxide - Organic & Biomolecular Chemistry (RSC Publishing)

Benzyl alcohol Benzyl group Benzylamine Chemistry Phenyl group, benzene ring, angle, white png | PNGEgg
Welcome to Chem Zipper.com......: Phenyl group is known to extract negative inductive effect, but each phenyl ring in biphenyl is more reactive than benzene towards Electrophilic substation. Why?